Chemical Dominoes:
In all sciences - Chemistry, Physics, Biology - there are always commonalities. For example, the human heart is an engineering masterpiece run by the pumping of blood from the heart. Plants are able to receive energy through photosynthesis, which involves the chemical reaction of sunlight, carbon dioxide, and water converting into glucose and oxygen. Each are intertwined with the other creating an ephemeral field consisting of new discoveries almost every day. In this project, we combined physics and chemistry concepts to create the Rube Goldberg of chemistry - chemical dominoes.
In all sciences - Chemistry, Physics, Biology - there are always commonalities. For example, the human heart is an engineering masterpiece run by the pumping of blood from the heart. Plants are able to receive energy through photosynthesis, which involves the chemical reaction of sunlight, carbon dioxide, and water converting into glucose and oxygen. Each are intertwined with the other creating an ephemeral field consisting of new discoveries almost every day. In this project, we combined physics and chemistry concepts to create the Rube Goldberg of chemistry - chemical dominoes.
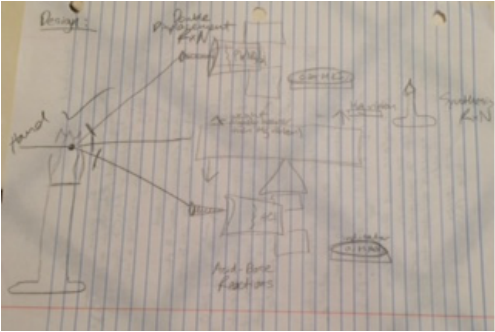
Blueprint:
Chemistry Concepts:
Below is the chemistry behind the four reactions in our chemical dominoes:
Below is the chemistry behind the four reactions in our chemical dominoes:
1. Combustion Reaction
Where: The match burns the convergence of the three ribbons holding the cups with various solutions.
Chemical Equation: C2H4(s) + 3O2(g) → 2CO2+ 2H2O(g)
Def: a substance combines with oxygen, releasing a large amount of energy in the form of light and heat. This produces carbon dioxide and water vapor
Where: The match burns the convergence of the three ribbons holding the cups with various solutions.
Chemical Equation: C2H4(s) + 3O2(g) → 2CO2+ 2H2O(g)
Def: a substance combines with oxygen, releasing a large amount of energy in the form of light and heat. This produces carbon dioxide and water vapor
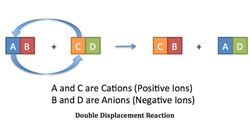
2. Double Displacement Reaction
Where: The lever drops lead nitrate into the beaker with potassium iodide. The solution turns yellow.
Chemical Formula: 2KI(aq)+ Pb(NO3)2 → 2K(NO3)2 + PbI2(s)
About:
Where: The lever drops lead nitrate into the beaker with potassium iodide. The solution turns yellow.
Chemical Formula: 2KI(aq)+ Pb(NO3)2 → 2K(NO3)2 + PbI2(s)
About:
- Double displacement reactions are extremely common.
- The yellow color appears when potassium reacts with nitrate.
3. Synthesis Reaction
Chemical Formula: 2Mg(s) + O2(g) → 2MgO(s)
Def: When two or more substances combine to form a new compound.
→ This is NOT a combustion reaction because the two substances form a new complex molecule, not carbon dioxide and water.
About:
Chemical Formula: 2Mg(s) + O2(g) → 2MgO(s)
Def: When two or more substances combine to form a new compound.
→ This is NOT a combustion reaction because the two substances form a new complex molecule, not carbon dioxide and water.
About:
- When the magnesium metal is exposed to atmospheric oxygen, it forms magnesium oxide, releasing a large amount of light and heat that is nearly impossible to stop once started.
- Magnesium metal is highly reactive.
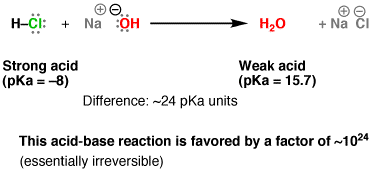
4. Acid-Base Reaction
Chemical Formula: NaOH(aq) + HCl(aq) → H2O(l) + NaCl(s)
Def: A chemical process in which hydrogen ions are exchanged between either neutral or electrically charged species.
About:
Chemical Formula: NaOH(aq) + HCl(aq) → H2O(l) + NaCl(s)
Def: A chemical process in which hydrogen ions are exchanged between either neutral or electrically charged species.
About:
- Neutralizes an acid or base in the solution
- Specifically to our reaction, the sodium hydroxide and the indicator form a blue color because it is basic. When the cup drops hydrochloric acid in the watch glass, the color of the solution changes to a light red color. The new solution, thanks to the indicator, can be identified as becoming acidic.
- These reactions are common in the body and atmosphere, like acid rain. Acid rain occurs when acidic gases are released by power plants and dropped many miles from their source, killing fish and plants.
- Antacids like Tums, Rolaids, and Maalox relieve heartburn by neutralizing stomach acid with a base.
- Acid-reducing medications like Pepcid AC, Tagamet HB, and Axid AR work by making the stomach secrete less acid.
Notes Involving Chemical Dominoes:
As mentioned before, before each lab, we took notes to help us understand the reactions being tested. Empiricism alone cannot teach class. Here are some of the concepts we covered:
As mentioned before, before each lab, we took notes to help us understand the reactions being tested. Empiricism alone cannot teach class. Here are some of the concepts we covered:
- Chemical Equations
- Describe a chemical reaction - the process by which one or more substances turns into one or more new substances; each substance has unique chemical and physical properties.
- Identify the reactants and products with each phase.
- When balanced, satisfy the Law of Conservation of Mass and show the relative amounts of all reactants and products.
- Ex: Photosynthesis --- 6CO2(g) + 6H2O(l) --> C6H12O6(s) + 6O2(g)
- Law of Conservation of Mass
- Matter cannot be created nor destroyed in a chemical reaction; only changed.
- In a chemical reaction, bonds are broken, atoms are rearranged, and new bonds are formed.
- The number and type of atoms on reactant side = the number and type of atoms on product side
- Mass of Reactants = Mass of Products
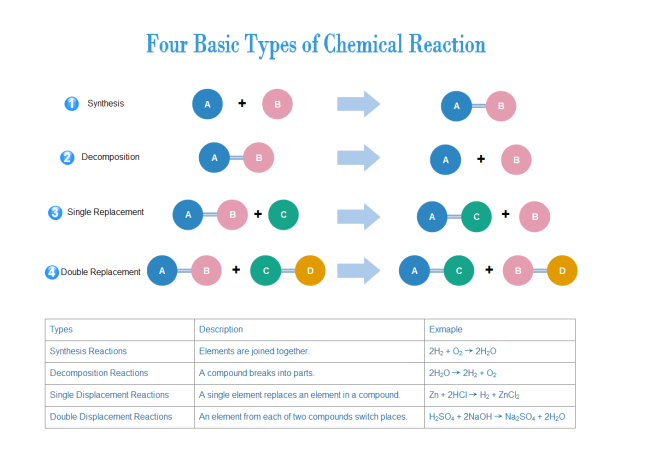
Types of Chemical Reactions:
- Synthesis - 2 or more reactants make one product
- Decomposition - one reactant yields 2 or more products
- Combustion of a Hydrocarbon - hydrogen or hydrocarbon burn in oxygen to water and carbon dioxide. Heat is given off as energy.
- Single Displacement - an element replaces its like an element in an ionic compound or acid.
- Double Displacement - one reactant replaces another part of the reactant.
Reflection:
With the involvement of corrosive chemicals and noxious flames, collaboration was key to the success of this project. Our group consisted of Jessica Walters, Zach Muzzarelli, Gabby Bitton, and myself. The beginnings of this assignment started with a series of labs and notes that contributed to the project. The notes would apply to the labs we would do on block days. Personally, I realized my lab skills needed improvement. For most of the labs, I was ready with full force to either inflate a balloon by heating up the substance in the balloon or place the magnesium ribbon over the Bunsen burner to create a blinding light. In fact, I was anxiously excited. However, patience was a virtue that was devoid from myself. Due to this unfortunate vice, I broke one Erlenmeyer flask, wasted 4.15 grams of lead nitrate, and many other little mistakes that slowed the lab progress down. In a few weeks, I am starting an internship at the Buck Institute. This lab helped me develop my lab skills, even at a much lower level.
After the background to the project was complete, our group began with the planning process. The blueprinting sessions commenced, yet the results seemed far from sight. Unlike the Rube Goldberg machines, a series of consecutive steps seemed more difficult with chemistry. Creating a new solution or lighting a bulb does not create a domino effect like a lever, pulley, or ramp does. Therefore, we found a solution. Instead, one piece of ribbon would be held over a match. At the convergent point, three ribbons would meet. The match would burn this uniting point due to combustion, releasing the three ribbons to initiate three more reactions. The next question was how can a loose ribbon start a reaction. Therefore, physics was essential to complete our chemical dominoes. Three levers would be attached to each ribbon: one dropping lead nitrate onto potassium iodide, one dropping hydrochloric acid onto sodium hydroxide and pH indicator, and one lower magnesium ribbon over a Bunsen burner to creating the ultraviolet rays. Now, we had to build the levers. If you have read my freshman year physics reflections, you'd know that I am, to say the least, insufficient with tools such as drills and saws. I wish I had been more helpful in assembling the levers instead of putting the labor on other members of my group. Also, using tools is useful for many other life activities. If I never try, I will never learn.
Two skills I improved on were my management skills and my chemistry knowledge. Management wise, I was able to do more of the hands on work instead of trying to take the "bossy" road. Usually, I'm overly concerned with finishing on time, but in this project, I discovered that quality beats punctuality most of the time, especially in chemistry. If we hadn't taken the time to test each reaction, the dominoes would lead to no effect like two clear solutions turning yellow or a purple solution turning pink. We did take longer to finish our project, however it worked perfectly for the video, without any human intervention. By collaborating as one mind instead of taking the leadership role, we were able to make the best project possible by combining our ideas, even if it did take longer.
What surprised me most about chemical dominoes was its "effect" on my understanding of chemistry concepts. More so than any previous units, I was able to grasp the concepts taught. For example, in action I saw how a double displacement reaction affected the characteristics of two solutions like turning yellow. The combustion reaction explained the effects of fire on flammable objects. When our lead nitrate proved to be inactive, we learned how to make a solution with the correct molarity of the substance using various conversion equations. This project truly expanded my knowledge of chemistry, at least in chemical reaction.
With every project, their comes the peaks and the pits, however insignificant or crucial they might be. One pit of the project started at the beginning when building the levers. Our first day of building was on a block day, so an hour and thirty-five minutes of work time. However, no one in our group thought to gather wood to work on. The bulk of the construction consisted of the levers, yet we were unable to work on them, giving us limited time to finish our project. Supplies are important to work on a project. A second pit was near the end of our project, which made it even more unfortunate. Our chemical dominoes started with a combustion reaction that led to three endpoints: the double displacement reaction, the acid-base reaction, and the synthesis reaction. Unknown to our group, the project must end with a bulb turning off due to silver nitrate corroding the wire. Because we had a successful and innovative design, we were an exception, but a full understanding of a project is vital to success in a project at school or at work in the future.
However, this project wasn't catastrophic. One of our peaks was when all our solutions worked to complete the needed chemical reactions. For some solutions, it took many trials to achieve the correct molarity but the desire results did come. The accuracy was crucial in order for our solutions to work. Another one of our peaks was the completion of our chemical dominoes. The camera was on, the levers were balanced correctly, the magnesium ribbon was directly above the Bunsen burner, and the ribbons had the correct tensions. The match was lit, releasing the three lever to create a yellow color from two clear liquids, neutralize the sodium hydroxide, and ignite the magnesium ribbon to create a bright light. This made the project worthwhile. To think, that one touch can initiate chemical dominoes.
With the involvement of corrosive chemicals and noxious flames, collaboration was key to the success of this project. Our group consisted of Jessica Walters, Zach Muzzarelli, Gabby Bitton, and myself. The beginnings of this assignment started with a series of labs and notes that contributed to the project. The notes would apply to the labs we would do on block days. Personally, I realized my lab skills needed improvement. For most of the labs, I was ready with full force to either inflate a balloon by heating up the substance in the balloon or place the magnesium ribbon over the Bunsen burner to create a blinding light. In fact, I was anxiously excited. However, patience was a virtue that was devoid from myself. Due to this unfortunate vice, I broke one Erlenmeyer flask, wasted 4.15 grams of lead nitrate, and many other little mistakes that slowed the lab progress down. In a few weeks, I am starting an internship at the Buck Institute. This lab helped me develop my lab skills, even at a much lower level.
After the background to the project was complete, our group began with the planning process. The blueprinting sessions commenced, yet the results seemed far from sight. Unlike the Rube Goldberg machines, a series of consecutive steps seemed more difficult with chemistry. Creating a new solution or lighting a bulb does not create a domino effect like a lever, pulley, or ramp does. Therefore, we found a solution. Instead, one piece of ribbon would be held over a match. At the convergent point, three ribbons would meet. The match would burn this uniting point due to combustion, releasing the three ribbons to initiate three more reactions. The next question was how can a loose ribbon start a reaction. Therefore, physics was essential to complete our chemical dominoes. Three levers would be attached to each ribbon: one dropping lead nitrate onto potassium iodide, one dropping hydrochloric acid onto sodium hydroxide and pH indicator, and one lower magnesium ribbon over a Bunsen burner to creating the ultraviolet rays. Now, we had to build the levers. If you have read my freshman year physics reflections, you'd know that I am, to say the least, insufficient with tools such as drills and saws. I wish I had been more helpful in assembling the levers instead of putting the labor on other members of my group. Also, using tools is useful for many other life activities. If I never try, I will never learn.
Two skills I improved on were my management skills and my chemistry knowledge. Management wise, I was able to do more of the hands on work instead of trying to take the "bossy" road. Usually, I'm overly concerned with finishing on time, but in this project, I discovered that quality beats punctuality most of the time, especially in chemistry. If we hadn't taken the time to test each reaction, the dominoes would lead to no effect like two clear solutions turning yellow or a purple solution turning pink. We did take longer to finish our project, however it worked perfectly for the video, without any human intervention. By collaborating as one mind instead of taking the leadership role, we were able to make the best project possible by combining our ideas, even if it did take longer.
What surprised me most about chemical dominoes was its "effect" on my understanding of chemistry concepts. More so than any previous units, I was able to grasp the concepts taught. For example, in action I saw how a double displacement reaction affected the characteristics of two solutions like turning yellow. The combustion reaction explained the effects of fire on flammable objects. When our lead nitrate proved to be inactive, we learned how to make a solution with the correct molarity of the substance using various conversion equations. This project truly expanded my knowledge of chemistry, at least in chemical reaction.
With every project, their comes the peaks and the pits, however insignificant or crucial they might be. One pit of the project started at the beginning when building the levers. Our first day of building was on a block day, so an hour and thirty-five minutes of work time. However, no one in our group thought to gather wood to work on. The bulk of the construction consisted of the levers, yet we were unable to work on them, giving us limited time to finish our project. Supplies are important to work on a project. A second pit was near the end of our project, which made it even more unfortunate. Our chemical dominoes started with a combustion reaction that led to three endpoints: the double displacement reaction, the acid-base reaction, and the synthesis reaction. Unknown to our group, the project must end with a bulb turning off due to silver nitrate corroding the wire. Because we had a successful and innovative design, we were an exception, but a full understanding of a project is vital to success in a project at school or at work in the future.
However, this project wasn't catastrophic. One of our peaks was when all our solutions worked to complete the needed chemical reactions. For some solutions, it took many trials to achieve the correct molarity but the desire results did come. The accuracy was crucial in order for our solutions to work. Another one of our peaks was the completion of our chemical dominoes. The camera was on, the levers were balanced correctly, the magnesium ribbon was directly above the Bunsen burner, and the ribbons had the correct tensions. The match was lit, releasing the three lever to create a yellow color from two clear liquids, neutralize the sodium hydroxide, and ignite the magnesium ribbon to create a bright light. This made the project worthwhile. To think, that one touch can initiate chemical dominoes.