RFP(Red Fluorescent Protein) Lab
Purpose: Make RFP from jellyfish in bacteria. Learn about the steps of genetic engineering.
Materials and Procedure:
Experimental Overview:
Part 2a: Verification of plasmid by restriction digest - cut plasmid with BamHI and Hind III to cut out RFP-ara from bacterial plasmid.
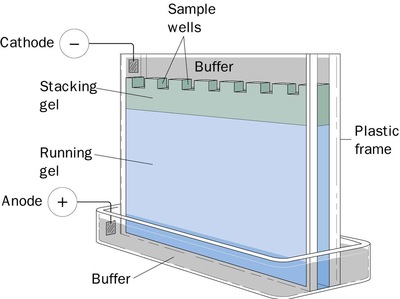
Part 4a: Verification of plasmid digest by electrophoresis.
Part 5a: Transformation of bacteria with recombinant plasmid.The relevant components on the plasmid are the red fluorescent protein (rfp) gene, the
promoter (pBAD), the ampicillin resistance gene (ampR), and the arabinose activator (araC).
Part 6: Portification of RFP using chromatography.
Results:
Before the 2a Lab:
1. If pARA-R is digested with BamHI and HindIII, what fragments are produced? Record the nucleotide sequence of the sticky ends and the length of each fragment (bp), and indicate the genes and other important sequences present on each fragment. Two fragments are produced. The two fragments are RFP with pBAD and Ara-C with ori with Amp-R. The RFP and pBAD is 807 BP, while Ara-C, ori, and Amp-R is 4495 BP.
2. In order to create a plasmid that can produce the red fluorescent protein in
bacteria, what components are needed in the plasmid? We need the RFP gene and Ara-C which binds to the promoter.
3. If the uptake of DNA by bacteria is inefficient (as discussed in the reading), why is a selectable marker (gene with resistance to an antibiotic) critical in cloning a gene in bacteria? The selectable marker is crucial because it determines the amount of bacteria that will grow and live while the other bacteria will die. This separates the bacteria into portions we need.
2a Questions:
1. List in words or indicate in a drawing the important features of a plasmid vector that are required to clone a gene. Explain the purpose of each feature. Ori = origin of replication; RFP = red fluorescent protein (gene of interest); Amp-R = selectable marker; Ara-C = binds to promoter so we get transcription of gene of interest.
2. What role do restriction enzymes have in nature? They are defense mechanisms. By cutting the DNA out of other bacteria, they die giving bacteria a better chance to retain nutrients from soil.
3. Using your understanding of evolution, why would bacteria retain a genethat gives them resistance to antibiotics? How is the existence of bacteriawith antibiotic resistance affecting medicine today?
This allows bacteria not to die off. If bacteria gains resistance, antibiotics won't work, killing us from diseases and illnesses that were harmful 300 years ago like an ear infection.
4. Bacteria, sea anemones, and humans seem, on the surface, to be very different organisms. Explain how a gene from humans or a sea anemone can be expressed in bacteria to make a product never before made in bacteria. All follow the same central dogma DNA to mRNA through RNA polymerase, and mRNA to a protein through ribosomes.
5. Due to a mishap in the lab, bacteria carrying a plasmid with an ampicillinresistant gene and bacteria carrying a plasmid with a gene that provides resistance to another antibiotic (kanamycin) were accidentally mixed together. Design an experiment that will allow you to sort out the two kinds of bacteria. Put Amp and Kan into a petri dish resistant to one. Repeat in another petri dish resistant to the other one. Once they are separated, we put them in different test tubes.
Purpose: Make RFP from jellyfish in bacteria. Learn about the steps of genetic engineering.
Materials and Procedure:
- 2a - materials and procedure in Amgen lab manual part 2a
- 4a - materials and procedure in Amgen lab manual part 4a
- 5a - materials and procedure in Amgen lab manual part 5a
- 6 - materials and procedure in Amgen lab manual part 6
Experimental Overview:
Part 2a: Verification of plasmid by restriction digest - cut plasmid with BamHI and Hind III to cut out RFP-ara from bacterial plasmid.
Part 4a: Verification of plasmid digest by electrophoresis.
Part 5a: Transformation of bacteria with recombinant plasmid.The relevant components on the plasmid are the red fluorescent protein (rfp) gene, the
promoter (pBAD), the ampicillin resistance gene (ampR), and the arabinose activator (araC).
Part 6: Portification of RFP using chromatography.
Results:
Before the 2a Lab:
1. If pARA-R is digested with BamHI and HindIII, what fragments are produced? Record the nucleotide sequence of the sticky ends and the length of each fragment (bp), and indicate the genes and other important sequences present on each fragment. Two fragments are produced. The two fragments are RFP with pBAD and Ara-C with ori with Amp-R. The RFP and pBAD is 807 BP, while Ara-C, ori, and Amp-R is 4495 BP.
2. In order to create a plasmid that can produce the red fluorescent protein in
bacteria, what components are needed in the plasmid? We need the RFP gene and Ara-C which binds to the promoter.
3. If the uptake of DNA by bacteria is inefficient (as discussed in the reading), why is a selectable marker (gene with resistance to an antibiotic) critical in cloning a gene in bacteria? The selectable marker is crucial because it determines the amount of bacteria that will grow and live while the other bacteria will die. This separates the bacteria into portions we need.
2a Questions:
1. List in words or indicate in a drawing the important features of a plasmid vector that are required to clone a gene. Explain the purpose of each feature. Ori = origin of replication; RFP = red fluorescent protein (gene of interest); Amp-R = selectable marker; Ara-C = binds to promoter so we get transcription of gene of interest.
2. What role do restriction enzymes have in nature? They are defense mechanisms. By cutting the DNA out of other bacteria, they die giving bacteria a better chance to retain nutrients from soil.
3. Using your understanding of evolution, why would bacteria retain a genethat gives them resistance to antibiotics? How is the existence of bacteriawith antibiotic resistance affecting medicine today?
This allows bacteria not to die off. If bacteria gains resistance, antibiotics won't work, killing us from diseases and illnesses that were harmful 300 years ago like an ear infection.
4. Bacteria, sea anemones, and humans seem, on the surface, to be very different organisms. Explain how a gene from humans or a sea anemone can be expressed in bacteria to make a product never before made in bacteria. All follow the same central dogma DNA to mRNA through RNA polymerase, and mRNA to a protein through ribosomes.
5. Due to a mishap in the lab, bacteria carrying a plasmid with an ampicillinresistant gene and bacteria carrying a plasmid with a gene that provides resistance to another antibiotic (kanamycin) were accidentally mixed together. Design an experiment that will allow you to sort out the two kinds of bacteria. Put Amp and Kan into a petri dish resistant to one. Repeat in another petri dish resistant to the other one. Once they are separated, we put them in different test tubes.
4a Questions:
1. The purpose of growing bacteria with ampicillin is that they are resistant and will not die. The p-Ara-R plasmid controls the cell's growth. Ampicillin kills unwanted bacteria.
2. You cannot control the production of your protein of interest since transcription cannot occur when the promoter isn't turned on by arabinose. RFP could not be expressed (no red).
3. I predicted the P+ in the LB plate will grow most abundant since it is the most inhabitual environment and P+ contains competent cells which support bacteria growth. Only cells with plasmids will grow.
5a Questions:
1. Our predictions were correct. The LB plate grew the most on it. The bacteria grew on both sides of the amp-ara, and on the LB-Amp, bacteria only grew on the P+ side but not the P- side.
2. There were no red colonies present either due to temperature or not enough time in the incubator.
3. The LB-amp-ara plate inhibits transcription to occur, which allows RFP to be expressed. The LB-amp does not have ara, a sugar.
4. It is important to have multiple copies because you can have extras for cell growth. Also, you can make more proteins.
5. The RFP gene is expressed as a trait through transcription from DNA to mRNA to protein (central dogma). This can happen because of ara-C.
6. Bacteria can again use transcription in the central dogma, and it can become a protein since it is resistant to different viruses, diseases, etc. Bacteria can make protein like humans because we both go through the same transcription.
6a Questions:
1. The red fluorescent protein can be depicted in each separation because the red cells will be at the bottom.
2. The supernatant was a clear tinted red liquid. The pellet is pink. The content was 150 microliters of EB and 150 microliters of LyB, along with red cells and bacteria.
6b Questions:
1. Binding Buffer (BB): create condition where amino acid and protein bind to the resin beads.
Wash Buffer (WB): washes off any proteins that are not tightly bound to the resin beads.
Elution Buffer (EB): high salt buffer, knocks protein off resin beads.
Column Equilibration Buffer (CEB): nice condition of salt and pH to store resin beads.
2. The supernatant was pinker than the last time. The pellet was only slightly darker pink than the supernatant. The content was 150 microliters of EB and 150 microliters of LyB, along with red cells and bacteria.
Lab 6 Observations:
Analysis and Conclusion: Many huge biotech companies use the Red Fluorescent Protein Lab in their work. For example, at BioMarin, they cut, paste, and copy DNA to make it beneficial. For example, we could use insulin in the recombinant DNA to help victims. In this lab, the red fluorescent protein represented the gene of interest like insulin. We could clearly see each step with the red fluorescent protein from the cutting with the restriction enzyme to chromatography.
Reflection: In this lab, Dr. L.B. picked the groups for us. My group was E.C. Schneider and Andrew Uyesugi. Our group was very efficient and cooperative with each other. Each person had a certain set of skills that made them proficient at a specific job. For example, E.C. was great at pipetting. Andrew was good at the mechanical jobs like the centrifuge and chromatography. I would read over the instructions and the questions first to decide the direction our group should be working towards. With a productive group, I truly enjoyed this lab. We were working with microscopic substances, but we could still easily see the results with the red fluorescent proteins. I enjoyed learning how this is incorporating in medical and pharmaceutical sciences. If I were to change anything about this lab, it would be to complete it over a shorter period of time. This lab took us over a month because we had the forensics project and the Lawrence Hall of Science accidentally sent us faulty materials (the protein wasn't red or fluorescent). Because we took so many breaks, I kept forgetting why and how we were doing this lab. All in all, the RFP Lab has been my favorite lab this year.
1. The purpose of growing bacteria with ampicillin is that they are resistant and will not die. The p-Ara-R plasmid controls the cell's growth. Ampicillin kills unwanted bacteria.
2. You cannot control the production of your protein of interest since transcription cannot occur when the promoter isn't turned on by arabinose. RFP could not be expressed (no red).
3. I predicted the P+ in the LB plate will grow most abundant since it is the most inhabitual environment and P+ contains competent cells which support bacteria growth. Only cells with plasmids will grow.
5a Questions:
1. Our predictions were correct. The LB plate grew the most on it. The bacteria grew on both sides of the amp-ara, and on the LB-Amp, bacteria only grew on the P+ side but not the P- side.
2. There were no red colonies present either due to temperature or not enough time in the incubator.
3. The LB-amp-ara plate inhibits transcription to occur, which allows RFP to be expressed. The LB-amp does not have ara, a sugar.
4. It is important to have multiple copies because you can have extras for cell growth. Also, you can make more proteins.
5. The RFP gene is expressed as a trait through transcription from DNA to mRNA to protein (central dogma). This can happen because of ara-C.
6. Bacteria can again use transcription in the central dogma, and it can become a protein since it is resistant to different viruses, diseases, etc. Bacteria can make protein like humans because we both go through the same transcription.
6a Questions:
1. The red fluorescent protein can be depicted in each separation because the red cells will be at the bottom.
2. The supernatant was a clear tinted red liquid. The pellet is pink. The content was 150 microliters of EB and 150 microliters of LyB, along with red cells and bacteria.
6b Questions:
1. Binding Buffer (BB): create condition where amino acid and protein bind to the resin beads.
Wash Buffer (WB): washes off any proteins that are not tightly bound to the resin beads.
Elution Buffer (EB): high salt buffer, knocks protein off resin beads.
Column Equilibration Buffer (CEB): nice condition of salt and pH to store resin beads.
2. The supernatant was pinker than the last time. The pellet was only slightly darker pink than the supernatant. The content was 150 microliters of EB and 150 microliters of LyB, along with red cells and bacteria.
Lab 6 Observations:
- After step 11, there was only one band at the top of the resin bed of red fluorescent protein.
- After step 14, the red fluorescent protein was spread throughout the top half of the resin bed.
- At step 16, the red fluorescent protein was a layer intertwined in the resin bed.
- After step 20, our red fluorescent protein after chromatography was not as concentrated and dark as others.
Analysis and Conclusion: Many huge biotech companies use the Red Fluorescent Protein Lab in their work. For example, at BioMarin, they cut, paste, and copy DNA to make it beneficial. For example, we could use insulin in the recombinant DNA to help victims. In this lab, the red fluorescent protein represented the gene of interest like insulin. We could clearly see each step with the red fluorescent protein from the cutting with the restriction enzyme to chromatography.
Reflection: In this lab, Dr. L.B. picked the groups for us. My group was E.C. Schneider and Andrew Uyesugi. Our group was very efficient and cooperative with each other. Each person had a certain set of skills that made them proficient at a specific job. For example, E.C. was great at pipetting. Andrew was good at the mechanical jobs like the centrifuge and chromatography. I would read over the instructions and the questions first to decide the direction our group should be working towards. With a productive group, I truly enjoyed this lab. We were working with microscopic substances, but we could still easily see the results with the red fluorescent proteins. I enjoyed learning how this is incorporating in medical and pharmaceutical sciences. If I were to change anything about this lab, it would be to complete it over a shorter period of time. This lab took us over a month because we had the forensics project and the Lawrence Hall of Science accidentally sent us faulty materials (the protein wasn't red or fluorescent). Because we took so many breaks, I kept forgetting why and how we were doing this lab. All in all, the RFP Lab has been my favorite lab this year.
Here is a powerpoint from the biotech company Amgen that explains what we did in this lab:
Here are some links to informational websites about Recombinant DNA and the manipulation of it:
http://education-portal.com/academy/lesson/restriction-enzymes-function-and-definition.html
http://education-portal.com/academy/lesson/what-is-a-dna-plasmid-importance-to-genetic-engineering.html
http://www.rpi.edu/dept/chem-eng/Biotech-Environ/Projects00/rdna/rdna.html
http://web.mit.edu/hst.160/www/quiz/RecombinantDNAAndCloning.htm
http://www.ncbi.nlm.nih.gov/books/NBK21881/
http://education-portal.com/academy/lesson/restriction-enzymes-function-and-definition.html
http://education-portal.com/academy/lesson/what-is-a-dna-plasmid-importance-to-genetic-engineering.html
http://www.rpi.edu/dept/chem-eng/Biotech-Environ/Projects00/rdna/rdna.html
http://web.mit.edu/hst.160/www/quiz/RecombinantDNAAndCloning.htm
http://www.ncbi.nlm.nih.gov/books/NBK21881/
RFP Gel Electrophoresis: